|
|
|
|

|

|

|

|

|

|

|

|
|
|
|
|

|

|

|

|

|

|

|

|
Sam Starcevic, John Hyatt, Xiaobo Hu, L.A. Lyon, Alberto Fernandez-Nieves
My
name is Sam Starcevic and I am a Research Assistant in the Soft Condensed
Matter Laboratory at Georgia Tech.Ā I
work under a Ph.D. student, John Hyatt, and Professor Alberto Fernandez-Nieves,
who heads the lab.Ā The following
highlights the work I have done while working with John.Ā There are other aspects of this overarching
project that are not detailed, as well as other projects that I am not a part
of that John works on.Ā If you wish to
learn more about them, please visit John HyattÆs page on this website.
Phase
Behavior of pNIPAM-PEG-AAc Microgels Studied by Viscometry
and Oscillatory Rheology
The
size of colloidal particles can range from 10 to 1000 nanometers.Ā Microgels are
colloidal particles of crosslinked polymers that are
both swollen and suspended by a solvent.
Some microgels swell and deswell
depending upon ambient properties of the suspension.Ā Parameters such as temperature and pH are
among those that can affect the size of the microgel.Ā The capacity of a microgel
to well within a solvent, then deswell,
expelling the solvent, especially at biological conditions, has possible
applications in the medical field.Ā The
structure and properties, for example the softness, of the microgels
can be radically different at different levels of swollenness.Ā This allows for characteristics of the
particles at different softness, such as the glass transition, to be studied.
This
project is of particular interest because of the temperature at which pNIPAM-PEG-AAc microgels undergo a change from a deswollen
state to a swollen state.Ā This occurs
around 31-33oC, which is close to body temperature.Ā This coincidence makes mapping of the
behavior of the microgels important base research for
medical applications.Ā pNIPAM has a lower critical
solution temperature transition from hydrophilic to hydrophobic.Ā Additionally, the AAc
copolymerized with the pNIPAM further ionizes at
higher pH, drawing in counterions to neutralize the
charge and swelling the microgel more, with some intraparticle Coulombic repulsion
from the ions.Ā Our interest is in the
phase behavior of dense suspensions of these particles, as well as intraparticle structure, specifically
that of the crosslinker position, as they deswell.
One
critical step to characterize the particles involves viscometry.Ā To obtain a phase transition curve dependent
on temperature and pH, a range of suspensions were prepared with differing microgel weight percentages.Ā The dynamic viscosity is determined using an Ubbelohde viscometer (see picture below).Ā The Einstein-Batchelor
equation (see below) is used to relate the dynamic viscosity
to the volume fraction of the microgels in the
suspension.Ā It is worth noting that the
Einstein-Batchelor equation is a model for hard
spheres.Ā However, the microgels are hydrodynamically
opaque, which contributes to their likeness to hard spheres and allows the
Einstein-Batchelor equation to be used for their
analysis.Ā The kinematic viscosity is
measured from the viscometry.Ā The dynamic viscosity is then obtained from
this value, and used to determine the volume fraction of the particles in
suspension.Ā Volume fraction is the
proportion of space that a solute occupies within the total volume of the
solvent.Ā This volume fraction
corresponds to swollen or deswollen microgels.Ā Since the
Einstein-Batchelor equation is only valid for lower
volume fractions (![]() ~0.2), this project is focused on establishing a base line
phase behavior graph from which higher volume fraction suspension behaviors can
be extrapolated.
~0.2), this project is focused on establishing a base line
phase behavior graph from which higher volume fraction suspension behaviors can
be extrapolated.
![]()
Where η is the
dynamic viscosity of the suspension, ηo
is the dynamic viscosity of the solvent, and ![]() is
the volume fraction.
is
the volume fraction.
The Ubbelohde
Viscometer
is shown lying in front of a water bath setup used to control the temperature
of the suspension.
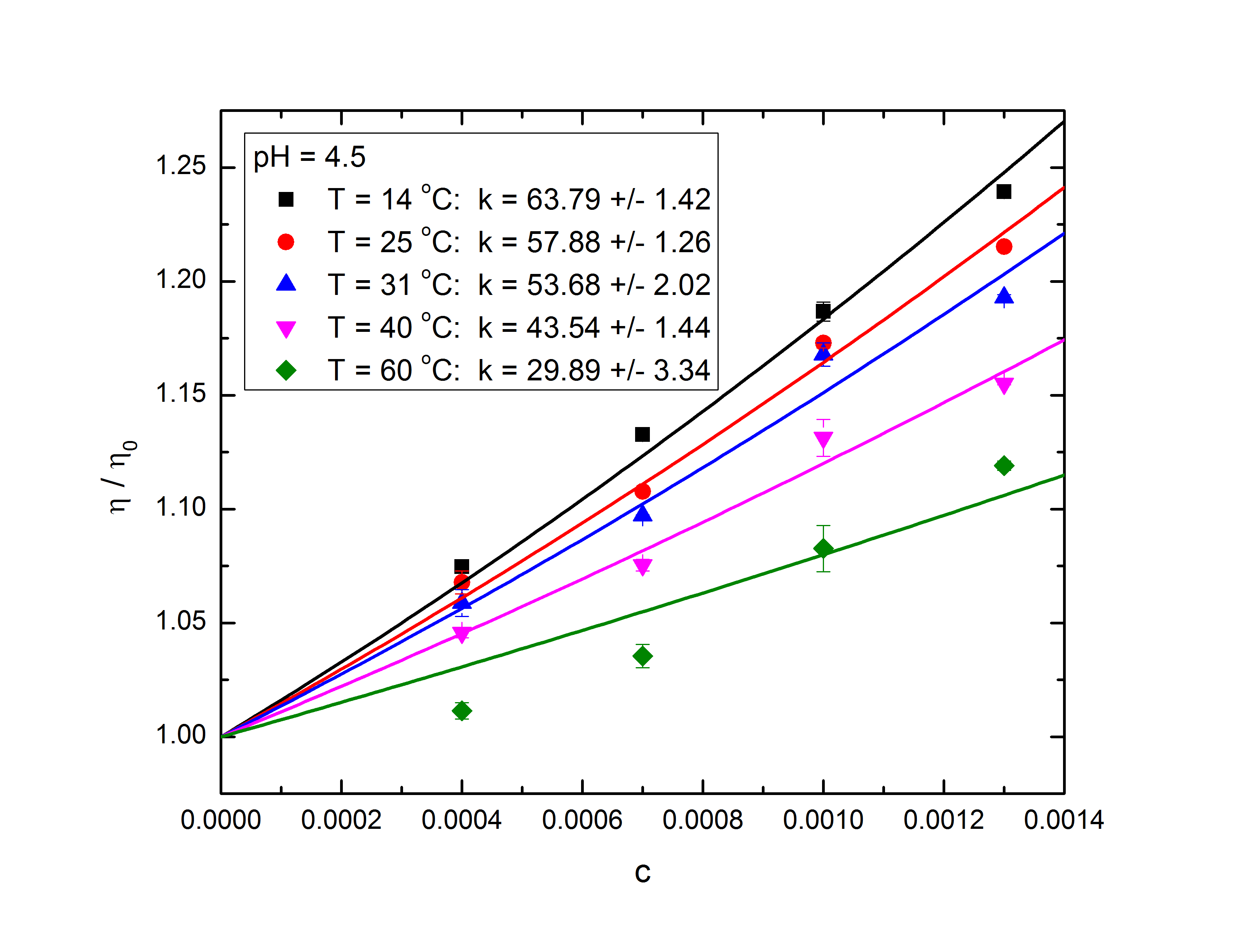
Some of our recent results from Viscometry.Ā The conditions of the experiment are a pH of
4.5 and a progression of temperature, separated by color in the graph.Ā Kinematic viscosity is obtained from the viscometry and converted to dynamic viscosity by
multiplying by the density of the solvent.
This is the value of ![]() Āin the Einstein Batchelor
equation.Ā This is divided by the
viscosity of the solvent (water) to obtain the ratio
Āin the Einstein Batchelor
equation.Ā This is divided by the
viscosity of the solvent (water) to obtain the ratio ![]() .Ā This value is shown above plotted against the
concentration of the microgel in the suspension.Ā A parabolic fit is applied to the data.Ā The coefficients,
.Ā This value is shown above plotted against the
concentration of the microgel in the suspension.Ā A parabolic fit is applied to the data.Ā The coefficients, ![]() Āand
Āand ![]() ,
correspond to kc
and k2c2, where c is
the concentration of the microgel in the suspension
and k is the
coefficient relating concentration and volume fraction.Ā The aim of this process is to eventually have
a graph of k dependent on both pH and
temperature for extrapolation and prediction purposes.
,
correspond to kc
and k2c2, where c is
the concentration of the microgel in the suspension
and k is the
coefficient relating concentration and volume fraction.Ā The aim of this process is to eventually have
a graph of k dependent on both pH and
temperature for extrapolation and prediction purposes.
The
second portion of the project utilizes the viscoelastic properties of the
suspensions that can be analyzed with rheology (see picture below).Ā The phase transitions of hard sphere
suspensions is much more studied and understood than that of microgels.Ā Microgels do not perfectly match the behavior of hard
spheres, but bare some resemblance to them, depending on how swollen they
are.Ā As they swell more, multiple microgels can occupy the same space, allowing for higher
effective volume fraction to be attained than with hard spheres.Ā This distinction differentiates the phase
transition of hard sphere suspensions and microgel
suspensions.Ā One area of interest to us
is the glass transition of the pNIPAM-PEG-AAc microgels.Ā The rhemoeter will
be used to study the storage modulus and loss modulus of suspensions of
different microgel weight percentage at various
temperatures and pH values.Ā This
information will be used to determine the behavior of the suspensions and the
volume fraction range that corresponds to the glass transition.Ā Additionally, the relaxation time of the
suspensions is of interest, which describes the time scale at which the microgels in suspension are able to flow around each other.
The Anton Paar Rheometer
Soft Condensed Matter Laboratory,
School of Physics, Georgia Institute of Technology
770 State Street NW, Atlanta, GA, 30332-0430, USA
Phone: 404-385-3667 Fax: 404-894-9958
sstarcevic3 [at] gatech.edu